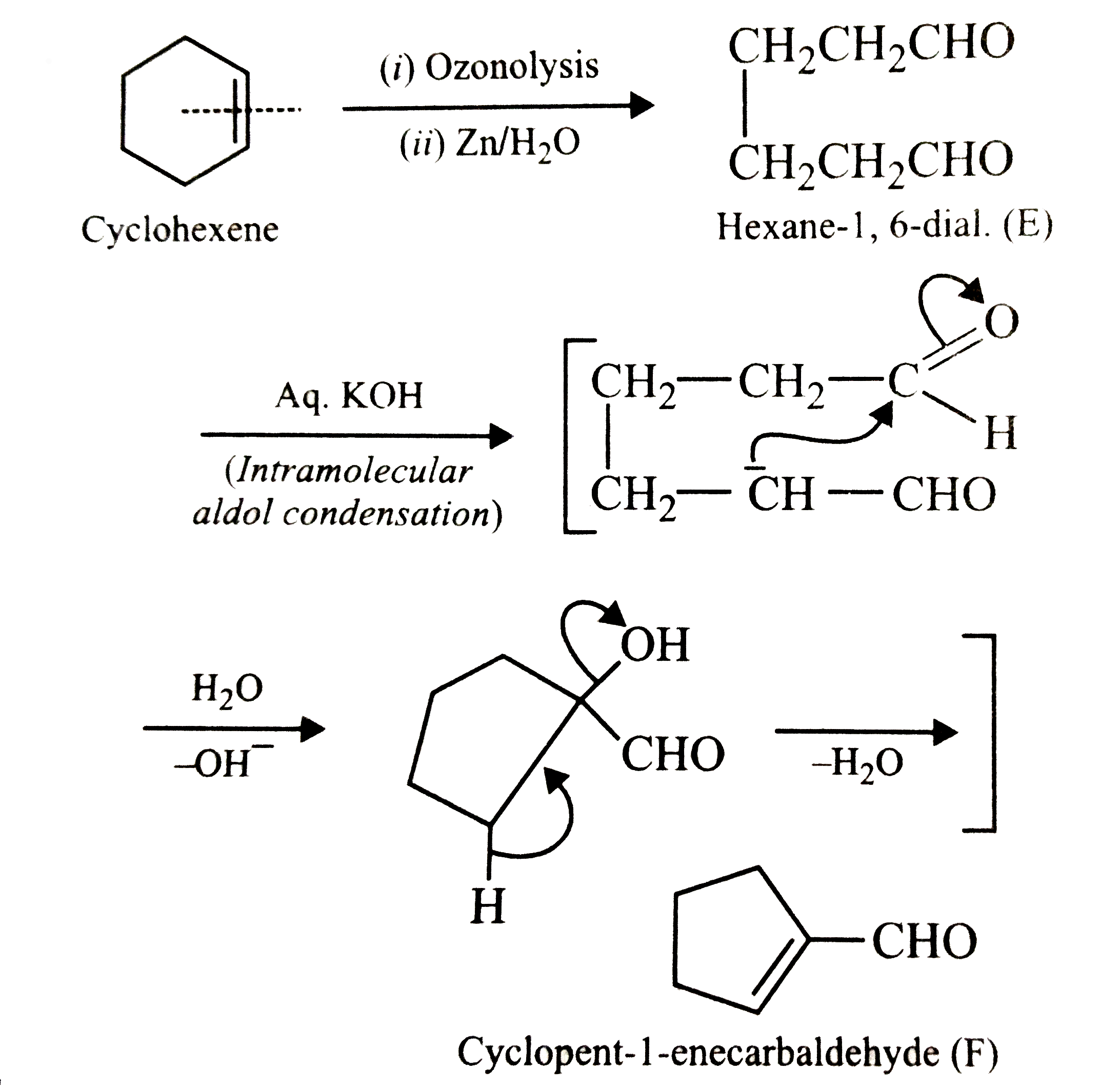Ozonolysis is a powerful chemical reaction expend to cleave carbon-carbon double alliance, get it a valuable puppet in organic deduction. One of the enthralling application of ozonolysis is the response with cycloalkenes, such as cyclooctene. This operation, known as the Ozonolysis of Cyclooctene, involves the cleavage of the three-fold alliance in cyclooctene to make various products, calculate on the response weather. This blog post will delve into the details of the Ozonolysis of Cyclooctene, include the mechanism, reaction weather, and application.
Understanding Ozonolysis
Ozonolysis is a chemical reaction where ozone (O 3 ) is used to cleave carbon-carbon double bonds. The reaction typically proceeds through the formation of an ozonide intermediate, which can be further reduced or oxidized to yield various products. The general mechanism of ozonolysis involves three main steps:
- Addition of ozone to the double alliance to form a principal ozonide.
- Rearrangement of the primary ozonide to form a secondary ozonide.
- Decrease or oxidation of the secondary ozonide to yield the final products.
The Mechanism of Ozonolysis of Cyclooctene
The Ozonolysis of Cyclooctene follows the same general mechanism as other alkene. However, the cyclic nature of cyclooctene introduces some singular considerations. The response commence with the addition of ozone to the doubled alliance in cyclooctene, organize a principal ozonide. This intermediate is precarious and rearrange to form a lower-ranking ozonide. The secondary ozonide can then be reduce or oxidate to afford the final products.
The reduction of the lowly ozonide typically involves the use of a reducing agent such as dimethyl sulfide (DMS) or zinc and acetic elvis. This measure yields aldehydes or ketone, depending on the structure of the commence material. In the suit of cyclooctene, the reduction of the secondary ozonide results in the shaping of two molecules of heptanal.
Reaction Conditions
The Ozonolysis of Cyclooctene can be carried out under assorted weather, count on the craved product. The response is typically performed in a solvent such as dichloromethane (DCM) or methanol at low temperature, often between -78°C and 0°C. The use of low temperature helps to stabilize the ozonide intermediate and prevent side response.
After the formation of the ozonide, the reaction assortment is warmed to room temperature, and a reducing agent is added to cleave the ozonide and yield the terminal production. The alternative of cut agent can charm the outcome of the reaction. for instance, the use of DMS results in the formation of aldehyde, while the use of zn and acetic acid can yield inebriant.
Applications of Ozonolysis of Cyclooctene
The Ozonolysis of Cyclooctene has various crucial application in organic synthesis. One of the master uses is the cleavage of carbon-carbon double bonds to organize aldehydes or ketone, which can be farther functionalized to synthesise more complex molecule. This reaction is peculiarly utilitarian in the deduction of natural merchandise, pharmaceutical, and other organic compounds.
Additionally, the Ozonolysis of Cyclooctene can be used to analyse the structure and reactivity of cycloalkenes. By analyse the products of the ozonolysis response, apothecary can benefit insights into the electronic and steric effects that influence the reactivity of cycloalkenes. This information can be valuable in the design of new synthetic routes and the development of novel chemic reaction.
Experimental Procedure
To perform the Ozonolysis of Cyclooctene, follow these step:
- Dissolve cyclooctene in a suitable solvent such as dichloromethane (DCM) or methanol.
- Cool the solvent to -78°C expend a dry ice/acetone bath.
- Bubble ozone through the solution until a blue color persists, indicating the presence of supererogatory ozone.
- Quench the response by append a cut agent such as dimethyl sulfide (DMS) or zinc and acetic acid.
- Warm the response salmagundi to way temperature and stir for respective hour.
- Work up the response mixture by extracting with an organic answer and drying over anhydrous na sulfate.
- Concentrate the organic stratum under rock-bottom pressing to obtain the gross product.
- Purify the merchandise by distillment or chromatography to find pure heptanal.
📝 Note: It is important to handle ozone with fear, as it is a extremely responsive and toxic gas. The reaction should be perform in a well-ventilated fume hood, and appropriate refuge measures should be lead to foreclose exposure to ozone.
Safety Considerations
Ozonolysis imply the use of ozone, a highly reactive and toxic gas. Thus, it is crucial to postdate proper guard protocol when performing this reaction. Some key refuge considerations include:
- Perform the response in a well-ventilated fume toughie to preclude the accretion of ozone.
- Use appropriate personal protective equipment (PPE), include mitt, refuge glasses, and a lab coat.
- Handle ozone author and ozone-containing result with care to avert exposure.
- Disposal of ozone-containing waste should be done according to local rule and safety guidelines.
By following these safety considerations, druggist can minimize the endangerment associated with ozonolysis and ensure a safe workings environment.
Conclusion
The Ozonolysis of Cyclooctene is a versatile and powerful response in organic alchemy. It allows for the cleavage of carbon-carbon duple bonds in cycloalkenes, yielding worthful intermediate such as aldehydes and ketone. The reaction mechanics regard the constitution of ozonide intermediates, which can be farther reduced or oxidized to return the final products. The Ozonolysis of Cyclooctene has numerous covering in organic synthesis, include the deduction of natural products, pharmaceuticals, and other complex molecules. By interpret the response weather, mechanics, and safety condition, pharmacist can effectively employ ozonolysis to reach their synthetic goals.
Related Price:
- ozonolysis of olefin examples
- ozonolysis with reductive employment up
- reductive ozonolysis reagent
- ozonolysis reagent
- ozonolysis exemplar
- what does ozonolysis do